The Center for Connected Health Policy (CCHP) has created this video primer on telehealth prescribing policy:
Rules for Prescribing Through Telehealth by Opioid Treatment Programs (OTPs)
- Historical Background: During the Public Health Emergency (PHE), the Substance Abuse and Mental Health Services Administration (SAMHSA) created a specific exemption for Opioid Treatment Programs (OTPs) from having to conduct an in-person physical evaluation of patients who were being treated with buprenorphine if an OTP physician, primary care physician or authorized healthcare professional under the supervision of the program physician determined that such an evaluation could take place via audio-visual or audio-only telehealth. SAMHSA’s flexibility was extended for one year after the end of the PHE (to May 2024) or until a final rule is published. On January 31, 2024, the Department of Health and Human Services (HHS) finalized rules for the prescribing of buprenorphine through the use of telehealth.
- The Final Rule: Consent for treatment can be obtained verbally or electronically and must be documented.
- Buprenorphine: Allows buprenorphine initiation at an OTP by the OTP practitioner if an OTP physician, primary care physician or other authorized health care professional under the supervision of a program physician determines that an adequate evaluation of the patient can be done via audio-visual or audio-only telehealth.
- Methadone: Audio-visual telehealth may be used “for any new patient who will be treated by the OTP with methadone if a program physician, or an authorized healthcare professional under the supervision of a program physician, determines that an adequate evaluation of the patient can be accomplished via an audio-visual telehealth platform. There is an exception to allow the use of audio-only to conduct the evaluation only when audio-visual is not available to the patient and the patient is with a licensed practitioner who is licensed to prescribe and dispense a controlled medication. The final rule is not applicable to, and does not authorize, the prescription of methadone pursuant to a telehealth visit.
The Ryan Haight Act and Prescribing of Controlled Substances
- Historical Background: Under the Ryan Haight Act of 2008, no controlled substance may be delivered, distributed or dispensed without a valid prescription. A valid prescription was one that was issued for legitimate medical purposes by 1) a practitioner who has conducted at least one in-person medical evaluation of the patient; or 2) a covering practitioner.
Prior to the COVID-19 pandemic and the declaration of the national Public Health Emergency (PHE), the Ryan Haight Act allowed for several narrowly tailored telemedicine exceptions to the in-person medical evaluation requirement. These included:
-
- If the patient is being treated in a DEA-registered facility (hospital or clinic)
- If the patient is being treated in the physical presence of another DEA registered practitioner
- If the telemedicine consult is conducted by a DEA registered practitioner for the Indian Health Service (IHS) and is designated as an Internet Eligible Controlled Substances Provider by the DEA
- If the telemedicine consult is conducted by a Veterans Health Administration (VHA) practitioner during a medical emergency recognized by the VHA
- During a public health emergency (PHE) declared by the Secretary of the US Dept. of Health and Human Services
- If the practitioner has obtained a DEA special registration for telemedicine or under other circumstances specified by future DEA regulations.
Neither a special registry nor a set of other circumstances has been established to date. In 2018, Congress passed the SUPPORT for Patients and Communities Act as part of an effort to combat the opioid epidemic. The Act directed the DEA to promulgate final regulations for the registry that would allow providers to prescribe controlled substances through telemedicine under certain circumstances. The DEA officially missed its deadline, set at one year from the passing of the Act on October 24, 2019.
- Impact of the Public Health Emergency (PHE). The declaration of a Public Health Emergency (PHE) in March 2020 triggered an allowance in federal law that temporarily allows for the expanded use of telemedicine in prescribing controlled substances for the duration of the PHE. Following is a decision tree summarizing the policy changes regarding How to Prescribe Controlled Substances to Patients During the COVID-19 Public Health Emergency
- What Happens When the PHE Ends? The allowances made for the PHE were set to expire at the end of the PHE (officially announced to end on May 11, 2023). However, the DEA has issued a temporary extension (read on to the very bottom for the new timeline)!
-
- In response to the lack of action by the DEA and given the worsening opioid overdose crisis, the Alliance for Connected Care convened more than 80 organizations, who jointly signed a letter urging the DEA to move forward with the telemedicine special registration process required by federal law that will enable SAMHSA waivered clinicians, community mental health centers and addiction treatment facilities to prescribe medication assisted treatment (MAT) drugs to patients with OUD employing telemedicine technology. To read about the issue in more detail and view the letter, see the Alliance for Connected Care’s webpage on the issue.
-
- In May 2021, Senator Warner sent a letter to Attorney General Merrick Garland regarding the long-delayed regulations and expressed great concern for the delay.
-
- In April 2022, Senators Portman and Whitehouse sent a letter to Administrator Anne Milgram of the U.S. Drug Enforcement Administration (DEA) and Secretary Xavier Becerra of the Department of Health and Human Services (HHS), urging the agencies to use their authority under the Ryan Haight Act to ensure Americans can continue to access important medication, such as Medication Assisted Therapies (MAT) for substance use disorder and other necessary drugs, via telehealth once the COVID-19 PHE ends.
-
- On February 24, 2023 the DEA announced proposed rules for permanent telemedicine flexibilities. While the proposed rule did include a few additional flexibilities, it did not include a mechanisms for implementing a provider registry. The Center for Connected Health Policy (CCHP) has created this video about the proposed rules:
-
-
- Read Press Release
- Read Proposed Rule
- See Proposed Rule Summary for Patients
- Following is a Summary Chart of the Proposed Rule:
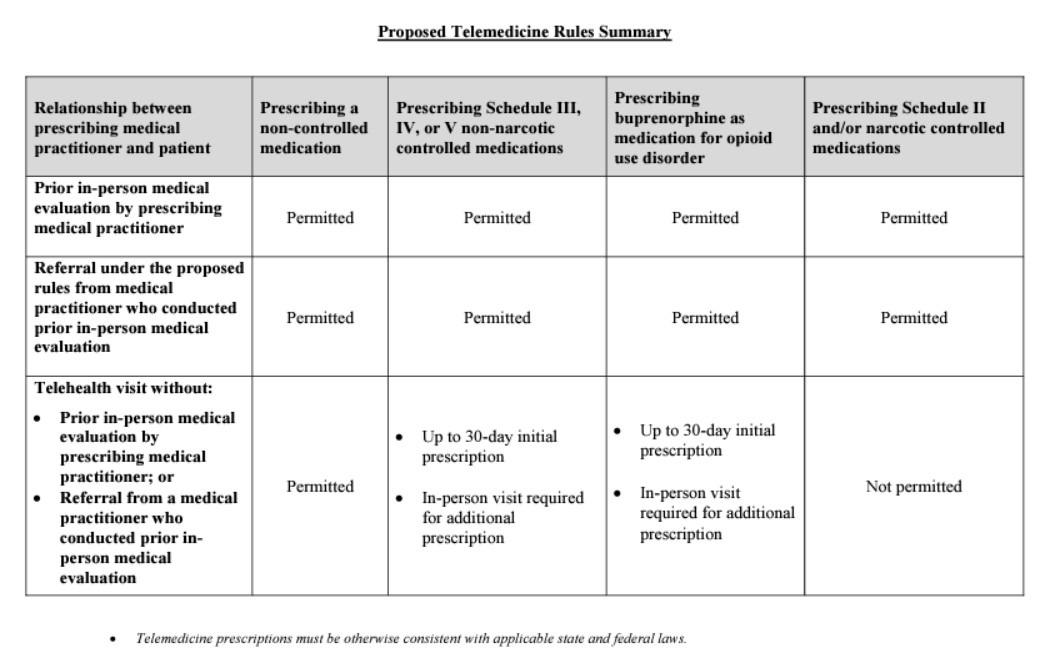
-
A 30-day public comment period about the proposed rule recently ended and comments were many! Here is a small sampling of the types of comments that were submitted:
-
-
- Read comments by attorneys from Foley & Lardner's Telemedicine & Digital Health Industry Team
- Read draft comments by the Telehealth Equity Coalition
- Read comments from the Center for Telehealth & eHealth Law (CTeL)
- Read press release and comments by American Telemedicine Association Action (ATA Action).
-
The DEA received a record 38,369 public comments. At the beginning of May, the DEA filed a draft temporary rule with the OMB titled "Temporary Extension of COVID-19 Telemedicine Flexibilities for Prescription of Controlled Medications".
The proposed "temporary" rule was published in the Federal Register on May 9, 2023 and was developed jointly between the DEA and SAMHSA.
Highlights of the Proposed Temporary Rule:
-
-
- More time is needed to review all the comments and to educate providers, patients and pharmacists about any upcoming changes.
- The PHE flexibilities will be extended through November 11, 2023.
- Any practitioner - patient telemedicine relationships established on/before November 11, 2023 will be permitted to continue under the PHE flexibilities through November 11, 2024..
-
-
- On October 6, 2023, the DEA and HHS extended the current telemedicine flexibilities through December 31, 2024. See “Second Temporary Extension of COVID-19 Telemedicine Flexibilities for Prescription of Controlled Medications,” in the Federal Register for full text.
While federal laws and actions of the DEA dictate how telehealth may be used to prescribe controlled substances, state laws also impact prescribing of medications. Therefore, practitioners also need to pay close attention to state policies (which differ by state).

